However, the Committee noted the clinical specialists' testimonies that most STEMI and NSTEMI patients would receive PCI. The Committee noted comments from consultees and commentators questioning the generalisability of the PLATO trial to UK clinical practice because most of the patients presenting with ACS in the UK would receive medical therapy only whereas 21% of patients in the PLATO trial received medical therapy only. The Committee understood that the manufacturer had accounted for age in its analysis. The Committee noted that the manufacturer had performed a large substudy of quality of life based on EQ-5D scores, which indicated no difference in the quality of life between people taking ticagrelor plus aspirin and those taking clopidogrel plus aspirin.Ĥ.4 The Committee heard from the clinical specialists that in general the trial was representative of the population in the UK, although the trial had a younger population and a higher proportion of men than the population with ACS in the UK. The Committee considered the clinical evidence for ticagrelor plus aspirin compared with clopidogrel plus aspirin in the subgroups of patients that were specified in the scope (STEMI, NSTEMI and unstable angina) and noted that the test for interaction showed no statistical difference between the groups (p = 0.41), interpreting this as no difference in the effectiveness between treatments by clinical presentation of ACS. The Committee also noted the non-statistically significant increase in the incidence of stroke, in particular haemorrhagic stroke, in patients randomised to the ticagrelor group. The Committee also noted that if the components of the primary end point were considered individually, the reductions in myocardial infarction and death from vascular causes were statistically significant (16% and 21% respectively) for patients randomised to the ticagrelor plus aspirin group. The Committee noted that ticagrelor plus aspirin reduced the relative risk of myocardial infarction, stroke and death from vascular causes by 16% compared with clopidogrel plus aspirin. The Committee noted that the manufacturer based its submission on a large trial, PLATO, which compared ticagrelor plus aspirin with clopidogrel plus aspirin. The clinical specialists stated that in the UK it is unusual for a patient with STEMI to undergo CABG and that approximately 10% of patients with NSTEMI undergo CABG.Ĥ.3 The Committee considered the evidence for the clinical effectiveness of ticagrelor compared with clopidogrel. 
The Committee understood that of people in the UK with ACS, few have unstable angina and often do not need revascularisation, but do receive dual antiplatelet therapy with clopidogrel and aspirin. The Committee heard from the clinical specialists that in the UK most people with NSTEMI undergo PCI. 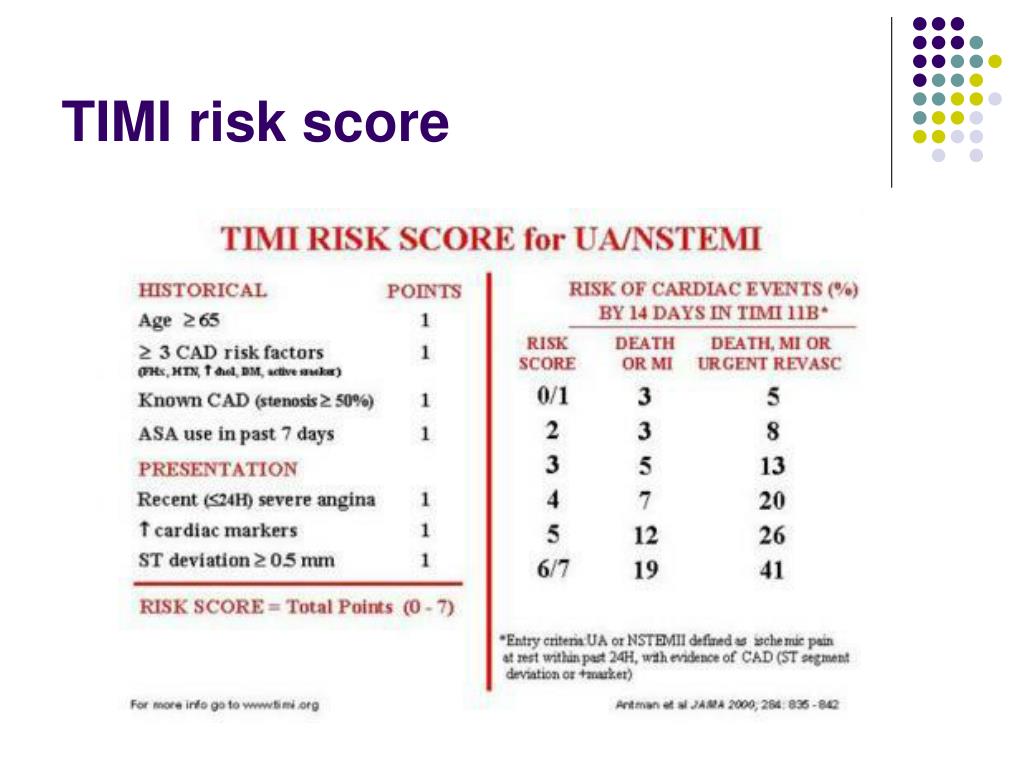
The Committee heard that in UK clinical practice people with NSTEMI are offered treatments depending on their GRACE or TIMI score medical management is an option for people at lowest risk of future adverse cardiovascular events, whereas people at higher risk would be offered PCI and subsequent dual antiplatelet therapy with clopidogrel and aspirin. The Committee heard that the duration of treatment of clopidogrel does not vary whether a stent is bare-metal or drug-eluting, because all people with ACS who undergo PCI, in the acute setting, are treated with clopidogrel plus aspirin for 12 months. It heard from the clinical specialists that, in the UK, treatment options for people with STEMI are prasugrel plus aspirin or clopidogrel plus aspirin, along with PCI with a bare-metal or drug-eluting stent followed by dual antiplatelet treatment. It also took into account the effective use of NHS resources.Ĥ.2 The Committee discussed the clinical management of ACS. 4.1 The Appraisal Committee reviewed the data available on the clinical and cost effectiveness of ticagrelor, having considered evidence on the nature of ACS and the value placed on the benefits of ticagrelor by people with these conditions, those who represent them, and clinical specialists.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
